Batteries are essential for powering our daily lives, from smartphones to electric cars. But why do batteries lose charge over time? This phenomenon is a common concern among users and manufacturers alike. Dr. Sarah Thompson, a leading expert in battery technology, highlights, "Every charge cycle impacts battery lifespan and efficiency."
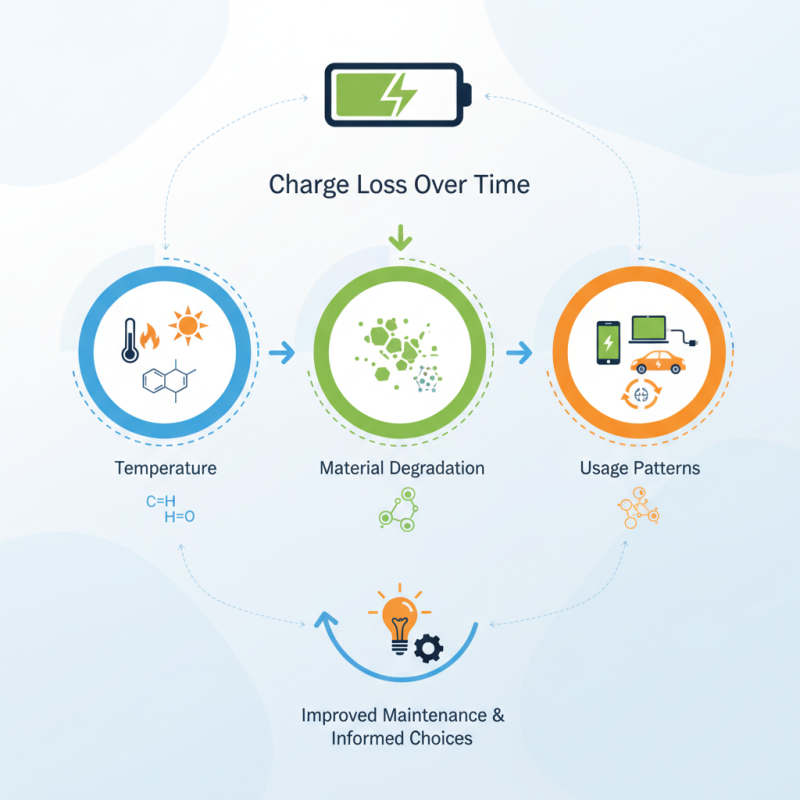
Understanding why batteries lose charge over time isn’t just a technical challenge. It's a crucial aspect of consumer experience. Factors such as temperature, material degradation, and usage patterns play significant roles. For instance, high temperatures can accelerate chemical reactions within batteries, causing faster deterioration.
The reality is that while battery technology has improved, it’s not perfect. Users often expect relentless performance, but the inherent limitations can lead to frustrating experiences. Recognizing these challenges is vital for better battery maintenance and usage practices. Embracing this knowledge can empower consumers to make informed choices about their devices.
Batteries generally lose charge due to various factors over time. Age is a significant element; older batteries naturally degrade. This degradation leads to reduced capacity, making them less efficient. Temperature also plays a vital role. High temperatures can cause accelerated chemical reactions. Conversely, extreme cold can slow these reactions, affecting performance.
Another key factor is the depth of discharge. Frequently draining a battery to its lowest point can shorten its lifespan. Keeping batteries partially charged can help maintain their health.
Tips: To extend your battery’s life, avoid extreme temperatures. It’s crucial to store batteries in a cool, dry location. Regularly check battery health, especially if it powers essential devices.
Ultimately, every decision impacts battery longevity. Choosing when to charge and discharge requires careful consideration. Reflect on how frequently you use your devices. Analyzing your habits can lead to more efficient battery maintenance.
Batteries lose charge over time due to complex chemical reactions. During discharge, electrochemical processes occur within the battery. In a typical battery, chemical compounds undergo changes, releasing energy. This energy powers our devices, but not without consequences.
One significant reaction involves the movement of ions. As the battery discharges, ions move from the anode to the cathode. This movement is essential for providing energy. However, with each cycle, these chemicals can degrade. Side reactions may produce unwanted byproducts. These byproducts can create resistance and diminish battery life.
Temperature and usage impact these chemical reactions as well. Higher temperatures can accelerate degradation. Conversely, extremely low temperatures could hinder reactions, leading to reduced performance. Users may not realize how their habits influence battery health.
Monitoring usage patterns could provide insights into maintaining optimal function, yet many overlook this. Reflecting on these factors can lead to a better understanding of how to care for batteries effectively.
Battery performance is significantly influenced by temperature. High temperatures accelerate chemical reactions within batteries. This can lead to faster degradation and reduced overall lifespan. A study by the Center for Advanced Life Cycle Engineering (CALCE) shows that battery life could decrease by up to 20% for every 10°C increase in operating temperature. Conversely, low temperatures slow down these reactions, leading to diminished capacity and efficiency, especially in lithium-ion batteries.
Proper temperature management is crucial for battery longevity. For instance, keeping batteries at optimal temperatures, typically between 20°C and 25°C, can enhance their performance. Excessive heat can result in thermal runaway, causing irreversible damage. Additionally, cold environments can reduce a battery's effective power. This understanding is vital for applications ranging from electric vehicles to consumer electronics.
Tips for battery care include: keep your batteries cool and avoid direct sunlight. Store devices in a temperature-controlled environment. Regularly monitor battery health and replace old units to maintain efficiency. Test your battery's performance under different temperatures to gauge its resilience. Awareness of these factors can help maximize battery life and performance in varied conditions.
Battery performance and longevity largely depend on their composition and design. Different materials used in battery production significantly impact how well they hold a charge over time. For example, lithium-ion batteries rely on a delicate balance of lithium, cobalt, and nickel. Each component plays a crucial role in charge retention. However, these materials can degrade, causing the battery to lose capacity quickly.
Design choices also affect charge retention. Thinner separators may increase energy density but can lead to faster degradation. The arrangement of electrodes and the choice of electrolyte can either enhance performance or lead to inefficiencies. For instance, battery packs that generate excessive heat during use can suffer from accelerated aging.
Moreover, we often overlook the mindsets behind battery design. Engineers strive for efficiency, yet some trade-offs may hinder longevity. It is essential to reflect on these decisions to improve future designs. Balancing performance and lifespan remains a complex challenge in battery technology. Understanding these factors can help consumers make informed choices.
Batteries gradually lose charge due to several factors. Temperature fluctuations, for instance, can impact battery performance. High heat can increase the rate of chemical reactions inside, leading to faster depletion. Conversely, extreme cold can hinder these reactions, reducing efficiency. Understanding these effects is crucial for extending battery life.
Implementing preventive measures can make a significant difference. Storing batteries in a temperature-controlled environment can help maintain their performance. Regular charging habits play a role too. Keeping batteries between 20% and 80% charge can prolong their lifespan. It's tempting to charge them fully or let them drain completely, but this may reveal weaknesses. Identifying your usage patterns allows for tailored strategies.
Moreover, periodic maintenance checks can identify degradation. Inspecting battery connections and cleanliness ensures optimal performance. While it may seem tedious, these small adjustments add up over time. Reflecting on your battery management choices is essential. Each decision can either contribute to longevity or accelerate wear. A little mindfulness goes a long way in battery care.
: Batteries undergo complex chemical reactions, releasing energy to power devices.
Ions move from the anode to the cathode during discharge, providing energy.
Chemical compounds can degrade over time, producing unwanted byproducts.
Higher temperatures speed up degradation, while very low temperatures can hinder performance.
Understanding usage patterns can help maintain optimal battery function and health.
Battery composition and design significantly affect how well batteries hold a charge.
Lithium, cobalt, and nickel are essential for maintaining charge retention.
Thinner separators may enhance energy density but lead to quicker degradation.
Balancing performance and lifespan is complex, often involving trade-offs.
Yes, understanding battery factors can help in making informed decisions.
Batteries are essential components in modern technology, but a common question arises: "Why do batteries lose charge over time?" Several factors contribute to this phenomenon, including chemical reactions that occur during discharge processes. As batteries provide energy, internal reactions can degrade the materials, leading to diminished capacity. Additionally, temperature plays a crucial role in battery performance and longevity; extreme heat or cold can accelerate charge loss and damage internal components.
The composition and design of batteries also significantly influence their ability to retain charge. Different materials and structural configurations can either enhance or reduce efficiency and lifespan. To mitigate charge depletion, it’s important to implement preventive measures such as optimal charging practices and temperature management. By understanding these elements, we can better appreciate the mechanisms behind battery charge loss and take steps to prolong their effective use.

 SK Battery
SK Battery